40 Years Old -- and Burning Down the House
Part 4 of a Series Discussing the Mechanisms behind the Non-Pharmaceutical Work-Arounds to COVID19 and Other Opportunistic Micro-Invaders, and to the Negative Effects of Aging
Introduction
As suggested at the beginning of this series, a silver lining of this whole COVID19 cultural, emotional, and economic digression is that some already established but neglected science is now being brought back up to the surface of human attention that, if it is applied more consistently and widely after the COVID19 furor, could eventually help alleviate a lot of present and future pain, suffering, and dysfunction -- especially within the older and/or chronically ailing members of the human standing crop. This is said because some of the therapeutic practices supported by this neglected science can, over the long run, help to curb some of the immuno-senescence and associated chronic inflammation brought on by disease, increasing age, suboptimal habits of diet and exercise, or environment. To provide some practical context, science shows that this process of developing immunosenescence and entering onto a course of chronic disease development begins to some degree for everyone by the age of 40-45. Some clear scientific evidence for this assertion regarding this transitional age will be related in more detail later.
This particular posting will relate some of the old and new science that is behind the COVID19-fighting approach of directly increasing the antioxidant capacity of the human body by supplementing the diet with such non-prescription substances as vitamins A, C, E, and melatonin, and indirectly doing so by supplementing with non-prescription N-acetylcysteine (NAC), vitamin D, and niacin. This discussion will pretty much finish up a consideration of all of the non-pharmaceutical, yet science-supported, approaches currently being brought to bear on COVID19.
At the end of this post, I provide evidence that geographically-determined, natural variation in one of the most important antioxidants made and used by the human body (and that of other animals as well) is responsible for a very considerable amount of the variation in US state level COVID19 mortality rate. Instead of saying, “Go west, young man, go west!”, this relationship could be referenced by saying, “Go north, old man, go north!”1
Burning down the House
Successful cellular invasion by COVID19 eventually leads to at least some amount of inflammation and cell death as immune system agents and processes do their work to try to stop and remove the viral invaders and any cellular damage that they may have caused. See Posting Figure 1 graphic below for a depiction of this process.2 In the worst cases, severe acute respiratory syndrome (“SARS”)3 develops, resulting in pneumonia with pulmonary alveolar collapse and possible sepsis and septic shock. Characteristically, utilization and inactivation of ACE2 receptor molecules by the COVID19/SARS-CoV-2 virus to gain cell entry notably helps accelerate and intensify this otherwise normal inflammatory housekeeping process to an unusual degree,4 increasing the probability of more severe illness in those with chronically belabored and therefore weakened immune systems.

Currently, the general approach to treating a moderate to severe cellular COVID19 infection is two-fold:5
Lower the viral load; and,
Reduce probability of development of sepsis so as to avoid multiple organ failure. Work towards this goal is chiefly conducted by controlling inflammation and inflammatory damage caused by an ‘overenthusiastic’ immune system response to the infection.
With moderate to severe infections, methods to reduce established and growing viral load variously consist of the therapeutic employment of antiviral Remdesivir, application of the zinc ionophores chloroquine or hydroxychloroquine, ivermectin, monoclonal antibodies, or Arbidol/Umifenovir.
Soto et al., 2020,6 explain and describe the general inflammatory process associated with a well-established COVID19 infection. This explanation and description should begin to provide insight as to why antioxidants are being used and/or recommended by some medical practitioners and scientists to help people with moderate to severe COVID19 infections minimize possible sepsis, and then recover from those infections:
In chronic obstructive pulmonary disease, acute lung injury (ALI) and acute respiratory distress syndrome (ARDS), there is an increase in reactive oxygen species (ROS) and reactive nitrogen species (RNS). These events are associated with the increased release of pro-inflammatory mediators7 such as IL-6, IL-8 and TNF-α by bronchial epithelial cells and alveolar macrophages, which may then activate neutrophils and macrophages, resulting in alveolar wall destruction and the collapse of small airways. These changes may induce endothelial damage, pulmonary capillary hyper-permeability and pulmonary edema, resulting in the deterioration of pulmonary gas exchange. Furthermore, in severe sepsis in ARDS, when there is life-threatening organ dysfunction caused by the host’s inadequate response to infection, the cardiovascular system increases the cardiac output and lowers peripheral resistance adopting a hemodynamic profile that leads to arterial dilatation. An excessive drop in peripheral resistance or its prolonged time course may result in progressive hypotension that is refractory to catecholamines and may contribute to severe cardiovascular failure. In different experimental models and in humans with severe septic shock, there is a high production and release of superoxide (O2−) and peroxynitrite (ONOO−) by different pathways that contri-butes to failure of the lungs, heart, brain and liver. Despite limited clinical data, moderate and severe septic shock is developed in many viral diseases such as SARS-CoV, which may increase the production of ROS and RNS. Overproduction of these molecules is associated with an elevated expression of iNOS, NADP oxidases, cyclooxygenase 2 and xanthine oxidase that activate transcription factors such NFkB resulting in an exacerbated proinflammatory host response. In addition, O2− and ONOO− participate as important mediators of the pro-inflammatory interleukin [cytokine] production. Moreover, these molecular species will continue to stimulate the production and release of more ROS and RNS which may interfere with mitochondrial respiration, since mitochondrial dysfunction is commonly induced in a septic shock environment.8
Putting it moderately more simply and much more generally (Gaschler and Stockwell, 2017):9
All biological systems exist in redox equilibrium, balancing oxidative and reducing reactions to achieve suitable conditions for life. Disruptions in redox homeostasis are caused by an accumulation of oxidizing molecules either by overproduction or loss of cellular reducing ability. In either case, the accumulated oxidizing agents are able to oxidize DNA, proteins, and lipids thereby altering their structure, activity, and physical properties. Given the potential severity of such widespread oxidative damage, perturbation of redox equilibrium can result in severe disruptions of biological homeostasis, potentially leading to disease or death.
Nevertheless, reactive oxygen species (ROS) and reactive nitrogen species (RNS) fulfill numerous critical and very necessary functions inside the human body. ROS as well as RNS are formed following normal cellular, tissue, and organ functioning. ROS results from the energy-producing oxidation that continuously occurs in the mitochondria and produces peroxides, superoxides, and hydroxyl radicals. RNS arise in part from the activity of immune cells such as macrophages, which (almost explosively) produce nitric oxide that reacts with superoxides to form peroxynitrite, a product that, while highly damaging to viruses and bacteria, also can inflict considerable collateral damage on the cell membranes, proteins, and DNA of the human body.
Cellular homeostasis and physical integrity can be maintained, however, by the sufficient presence of antioxidant enzymes10 like catalases, lactoperoxidase, superoxide dismutases, glutathione peroxidase, and antioxidant molecules like melatonin, glutathione, and vitamins A, C, and E.11 These molecular substances all function as backstops or fail-safes to the overproduction of reactive oxygen and nitrogen species, buffering the body’s redox systems so that little irrecoverable collateral damage to the body’s cells and tissues occurs when the body’s immune system has to go, for the greater good, into oxidizing ‘hyperdrive’ to defend the body against viral or bacterial invaders.
However, with continued extreme use of the body’s redox systems – or reduction in level of maintenance of these systems -- the backstop or fail-safe levels of glutathione and other antioxidants decrease under oxidative stress, and ROS and RNS begin freely damaging, breaking down -- and even killing -- the cells, tissues, and organs making up the body’s functional structures. This is what happens in the worst-case COVID19 virus infections especially. See Posting Figure 1 bottom left hand diagram again.
What and Where Matters with Antioxidants
According to John and Platts (2014),12 there are three basic locational categories of antioxidants based on whether they are chemically lipophilic, hydrophilic, or amphiphilic. Lipophilic antioxidants like vitamin A, beta-carotene, and vitamin E attach to, and protect, the lipid membranes encapsulating cells. Hydrophilic vitamin C and glutathione, on the other hand, mostly protect the constituents of aqueous intracellular environments of the cytoplasm and extracellular fluids like blood. Melatonin, on the other hand, is both lipophilic and hydrophilic (i.e., is amphiphilic), so is found everywhere in the body and indeed extremely rapidly crosses the semipermeable membranes of the blood-brain and placental barriers.
Again, according to John and Platts (ibid.):
Vitamin E or α-tocopherol has been described the “last line of defense” in a multicomponent endogenous antioxidant system. It appears that under conditions of stress, depletion of cellular ascorbic acid [vitamin C] occurs first, followed by glutathione, then α-tocopherol [vitamin E], [then] resulting in initiation of lipid peroxidation [and consequent cell damage and destruction].13 When glutathione is depleted, ascorbic acid plays a vital role in maintaining cellular α-tocopherol levels and survival of the cell. One might expect that melatonin should be depleted after α-tocopherol, particularly in membranes, as it is higher in the electrochemical series at 700 mV, compared to 500 mV for α-tocopherol. Melatonin may therefore truly be the last line of defense against oxidative damage.
Tan et al., 2006,14 however, provide evidence indicating that melatonin is also the first antioxidant to be employed by the body to cope with reactive species in cases where oxidative stress increases suddenly. This suggests that, strictly speaking, melatonin is actually both the first and last line of defense against oxidative damage in human bodies.
For the reference and convenience of people who remember general chemistry, the electrochemical series of some biochemically relevant antioxidants and other molecules is illustrated in Posting Figure 2 below. Note that the position of the reactive oxygen and nitrogen species on the far right of this diagram. These ‘right wing extremists’ can oxidize the molecules to their left, while the ‘left wing extremist’ antioxidant species can scavenge and ‘quench’ the reactive species on the right, protecting important cell, tissue, and organ structures from oxidative damage. Overall redox potential of the plasma of a healthy 25-35 year old healthy human is reported to be about -140 mV,15 which is at about the same redox potential as vitamin C.

Whereas all of the antioxidant molecules and enzymes -- besides melatonin and its several metabolites16 -- are recycled (reduced once again) after initially scavenging an oxygen or nitrogen reactive free radical, this recycling occurs only by transferring the extra electron(s) contained in the initially problematic oxidizing species to yet another antioxidant molecule in the system, thus only ‘kicking the can on down the road’ and using up some of the body’s energy production and stores in the meantime.17 In the worst case, this antioxidant recycling itself can cause structural damage to important body molecules. In contrast, a single molecule of melatonin, purportedly the first and last line of defense against oxidative damage of the body’s cells, tissues, and organs, is a ‘sacrificial’ antioxidant in that it will ultimately scavenge and quench up to ten different free radical electrons originating from ROS or RNS before terminally becoming exhausted of antioxidant capacity and disposed in the urine (Johns and Platts, 2014, ibid.). So, during the scavenging and antioxidant activity of melatonin, the harmful ROS or RNS is actually exported out of the body as waste, not just ‘kicked on down the road’ onto another hapless and innocent molecule making up part of the body.
Direct Antioxidant Treatment of COVID19 Infection
This last free radical disposal process by melatonin is significant as far as bodily oxidation stress and effective immunity level are concerned, inasmuch as daily melatonin production declines extremely markedly with increasing human age.18 This decline in daily melatonin production with age evidently gradually and progressively encumbers the entire ROS and RNS control process. It has a similar effect on innate and acquired immunity systems as well (ibid.). See Posting Figure 3 comparing daily melatonin production of different age classes of people showing that older people have much, much less of the critical melatonin antioxidant on hand to work with each day than younger people.19

The drop in daily melatonin production is paralleled by the decrease in glutathione (the other major endogenous antioxidant resource of the human body) that occurs in people with increasing age. Posting Figure 4 of plasma glutathione again indicates that, on average, people become progressively more antioxidant-poor and therefore more chronically inflamed and under more oxidative stress after the age of 40. On the other hand, these graph data also suggest that about 75% of the people who manage to live into and past their 80s are, by their individual nature, less chronically-inflamed and suffer less oxidative stress than those who fail to make it into their 80s and beyond.

The point is, older people, and people with chronic diseases like diabetes,20 athero-sclerosis,21 and the male gender (sic),22 exist at higher level of chronic inflammation and oxidative stress (OS), leaving them with less available antioxidant resources to handle acute inflammation and biomolecular oxidation episodes like those that can be suddenly brought on by infections like COVID19. See and compare the lower left- and right-hand diagrams in Posting Figure 1 again.
Accordingly, researchers and clinicians, motivated by the COVID19 event have searched the scientific literature for leads to clinical tools to help safely moderate the immediate and after effects of a COVID19 viral infection of the cells of people already under oxidative stress because of age or infirmity. A leading, early example of this work with antioxidants was carried out in Mexico during 2020. Because the very earliest reported Chinese COVID19 treatment experience23 indicated that anti-inflammatory steroid medicines delayed viral clearance and did not promote survival, Chavaria et al., 2/2021,24 instead tested well-known nonprescription antioxidants like melatonin, vitamin C, vitamin E, and N-acetylcysteine (“NAC”), to evaluate the effect of these inflammation-moderating antioxidants on the health outcomes of hospitalized patients suffering from moderate to severe COVID-19, with and without sepsis.
Five-day treatment dosages of the nutraceuticals used in their hospital study were 1000 mg vitamin C twice daily; 400 IU vitamin E (alpha-tocopherol acetate) twice daily; 600 mg N-acetylcysteine (NAC)25 twice daily; 50 mg timed-release melatonin26 twice daily. Each patient was also given, evidently as a precautionary measure, 400 mg of the prescription nonsteroidal anti-inflammatory, pentoxifylline, in addition to supplementation with one of the four named antioxidants.
Results were that oxidative stress markers were reduced and five different clinical measures of COVID19 infection survivability improved in all but 3 of the 110 study patients. Vitamin E and glutathione-increasing NAC worked best with the severely infected patients in reducing oxidative stress levels, while vitamin C and melatonin were most effective with patients with moderately severe COVID19. These improve-ment results with single antioxidants added to pentoxifylline exceeded those for the ‘control’ patients given prescription pentoxifylline only. Author-managers of the clinical study were of the opinion that combining the antioxidants into a multi-component antioxidant cocktail would have improved the clinical results further. This is extremely likely to be true, as previous research has demonstrated that the combined antioxidant operation of vitamin C, vitamin E, and melatonin is very highly synergistic.27
It is suspected that the Chavaria et al., (ibid.) clinical study results are the original source of the vitamin C and melatonin antioxidant recommendations promulgated to the public by FLCCC, as an example:

Indirect Anti-inflammatory Treatment of COVID19 Infection
Two additional substances have been shown to appreciably modulate the damaging inflammation caused by ROS and RNS released by human immune systems during, for example, COVID19 infections. The most commonly recognized and used supplement concerned is vitamin D, while a lesser known and currently less frequently applied substance is vitamin B3 (especially the nicotinic acid form of B3). Both of these vitamins do not serve commonly as antioxidants like vitamins C, E, or like melatonin or glutathione, to – after the fact of ROS or RNS generation -- scavenge and quench ROS and RNS. Instead, they reduce the negative effects of immune system release of ROS and RNS by acting ‘upstream’ of the immune system’s potentially damaging inflammatory activity to help regulate and moderate that activity before it has a chance to get out of hand and become overly damaging.
Vitamin D
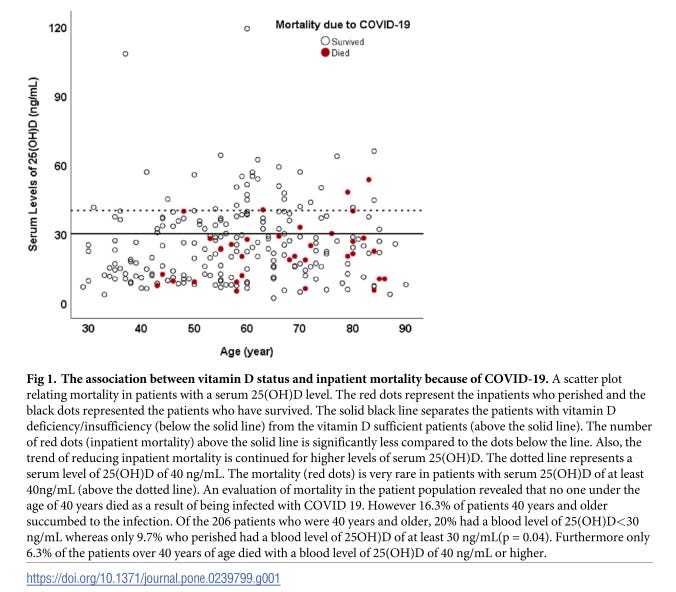
It is the prevailing working hypothesis28 that vitamin D attenuates the cytokine/-chemokine storm that can accelerate a COVID19 infection into Acute Respiratory Distress Syndrome (ARDS) and possible sepsis, reduces the pro-inflammatory effect of the COVID19 virus interference with ACE2 receptor molecules, modulates activity of ROS-secreting neutrophils, tightens cell junctions, stimulates repair of inflammation-damaged lung epithelium, and reduces probability of COVID19-related clotting problems. It is therefore not surprising that the scatter plot below, Posting Figure 6, empirically indicates there is a significant probability that avoiding vitamin D deficiency by getting regular sunshine exposure and/or supplementing sufficiently with the vitamin is important with regard to the goal of avoiding COVID19 mortality – especially for – yet once again -- people over the age of 40.29
Because the more fragile elderly and/or the chronically ill are much more likely to be housebound and therefore receive very little, if any, daily sunshine exposure, they are most likely to evidence vitamin D deficiency. In addition, the skin of older people produces as little as 75% less vitamin D from sun exposure.30 Old or not, average vitamin D sufficiency (>30 ng/mL) in the US Caucasian population declined from about 60% in 1988-1994 to 30% in 2001-2004. Over the same time period, average US African-American vitamin D sufficiency even more critically declined from 10% to 5% (ibid.).31 Suspected reasons for these overall declines include changes in cultural habits (e.g., less recreational and work-related outdoor activity, increasing obesity, and the new — and somewhat trendy and fashionable — widespread use of sunscreen ointments). Evidently, ordinary dietary vitamin D intake is not sufficient to compen-sate for changes in cultural practices serving to reduce vitamin D production from sun exposure, and from the bodily vitamin D ‘dilution’ caused by the increasing incidence of body mass-increasing obesity in the US.32 This makes vitamin D supplementation not only a concern for the housebound and elderly, but also for the sunscreen-slathered young, the obese, and/or strongly melanin-blessed.
Alam et al., 2021,33 besides providing a listing of the recent studies indicating the utility of vitamin D supplementation in reducing the chances of dying from a COVID19 infection, provide a Table detailing the amount of vitamin D supplementation required to help provide for the prevention and treatment of COVID19 (and infections from other respiratory pathogens) of people of various ages and health statuses:
NAD Supplementation – and Zinc Once Again!
NAD (nicotinamide adenine dinucleotide) is a very abundant and an absolutely biochemically critical coenzyme derived from the various chemical forms of vitamin B3 and the amino acid tryptophan that, among other things, shuttles electrons around the cytosol and mitochondria to produce all of the energy used by human (and other animal) cells, tissues, and organs. NAD accepts and donates electrons with almost equal facility, so functions readily as both an oxidizing agent and reducing agent.34 During this electron-shuttling work per se, no NAD is consumed and lost from further energy-generating use by the body – the NAD is just repeatedly reduced and oxidized as each human cell converts food sources to the energy form (ATP) it is uses for metabolism.35 However, besides serving as an electron-transferring coenzyme that is not consumed by the process of producing cellular energy, NAD is also consumed as a substrate by members of at least three different enzyme families:
The PARP (poly ADP-ribose polymerase) family of enzymes is involved in the repair of nuclear DNA, genomic stability, and programmed cell death. PARPS use NAD as a substrate during their enzyme activities and produce nicotinamide (NAM) from that NAD as a metabolic waste product. Upon sufficiently massive nuclear DNA damage by RNS or ROS, increasing PARP activity can consume 80-90% of the cellular NAD supply available in the mitochondria, and cell nucleus and cytoplasm. This NAD overconsumption shuts down cellular energy production, destroys the mitochondria, and kills the affected cell.
Sirtuins (NAD-consuming protein deacetylases).36 Seven different sirtuins (SIRTS) operate intercellularly, use NAD for an enzymatic substrate, and also produce NAM as a reaction waste product. Some sirtuins affect cell longevity and cell death. Others, like the PARPS discussed above, play a part in DNA repair and cell metabolism after cell damage (e.g., influence the course of inflammation processes). Sirtuin activity is turned off by elevation of waste product NAM accumulated in a cell, however. And, because PARPS utilize much more NAD and therefore produce more NAM than SIRTS in the event of oxidative or other damage of DNA, this means that SIRT activity can be turned on and off by the cellular level of PARP activity. Further, because SIRT1 in particular controls FOXO protein expression, only SIRT1’s continued activity under low NAM concentrations insures operation of FOXO-controlled genes responsible for cell cycle arrest, ROS scavenging and quenching, DNA damage repair, and inhibition of cell death. In addition to low environmental concentration of NAM, and high environmental concentration of NAD, the SIRT1 molecule also requires structural coordination with a divalent zinc cation (Zn 2+) to be functional. The combined and therefore active Zn-NAD-SIRT1 moieties minimize cytokine release and the consequent hyperinflammation like that associated with severe COVID19 infections.37 Recall here the earlier discussion of the demonstrated role zinc and its ionophores also play in maintaining innate immunity against COVID19.
CD38 (cyclic ADP-ribose hydrolase) is another enzyme/receptor that consumes especially large quantities of NAD. This enzyme molecule penetrates through the cell and nuclear membranes of macrophages, the cells of the heart, skeletal muscles, nerves, brain, pulmonary arteries, lymphoid system, lungs, digestive tract, and pancreas.38 Besides functioning as an activator of extracellular immune cells, CD38 also produces messenger molecules that cause cellular release of calcium – and production of much waste NAM. Tellingly, the CD38 enzyme becomes a more and more ubiquitous and active NAD consumer and producer of waste NAM in the cells of animal bodies as they age.39 It is suspected by most researchers in the field that the voracious NAD-consuming activity of CD38 is a major, if not the major, reason for declining NAD concentrations observed in aging human bodies.
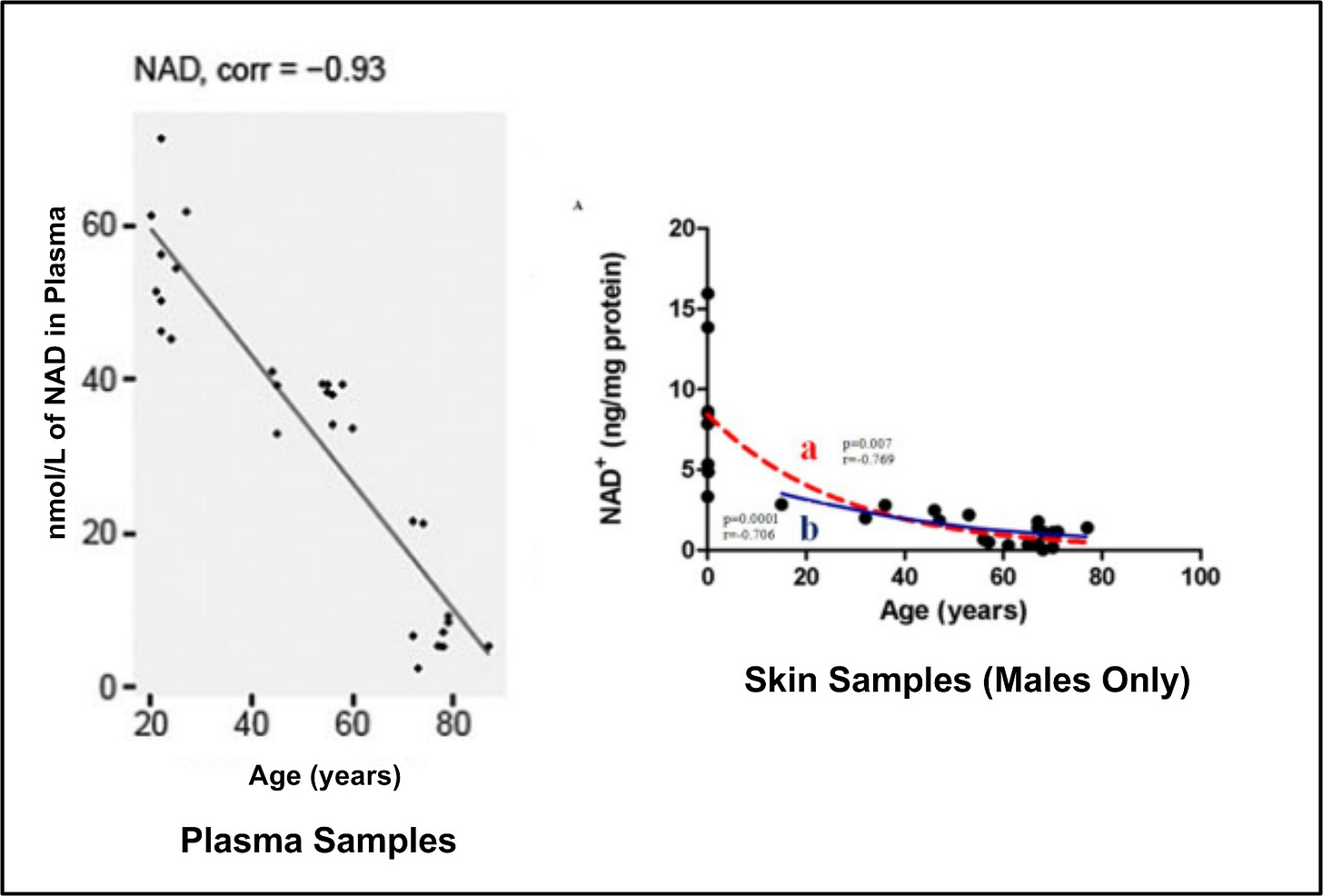
Over the long run, consequent of the increasing NAD-consuming activity of the enzymes described above40 under the influence of chronic oxidative stress and consequent cell (and DNA) damage and destruction, NAD levels in tissues and plasma decline continuously with increasing age and/or during the course of some diseases. See Posting Figure 7 below. This means that the old, and other people under illness-related (e.g., diabetes, obesity, hypertension, cardiac disease) oxidative stress, face COVID19 infection from an initial position of energetic weakness and low overall vitality because of chronic NAD depletion. Further, it has been determined that the COVID19 virus itself actively consumes intercellular NAD stores and hinders NAD replacement during virus infection and replication.41
Although NAD supplementation therapy has already been shown to correct many of the negative effects of aging,42 its use is prospective and theoretical at this time with regard to the problem of avoiding and fighting the complications of COVID19 infections. The potentially preventive and curative antiviral therapy is actively being tested43 with regard to COVID19, however.
Given that it has been very recently shown44 that oral NAD supplementation with the very expensive45 proprietary NAD precursors nicotinamide riboside (NR) and nicotinamide mononucleotide (NMN) effectively only results in the same augmentation of the body’s NAD stores as achieved with nonproprietary and inexpensive46 nicotinic acid (NA) and nicotinamide (NAM) NAD precursors, those interested in NAD supplementation may well want to use one of the two inexpensive, nonproprietary NAD precursors. In that regard, Pinen et al., 2020,47 showed in a landmark pilot clinical study that supplementation with 250 mg to 1000 mg per day of nicotinic acid quite rapidly rescued age-related cellular NAD and mitochondrial decline. See graphic abstract of the paper below in Posting Figure 8.

Finally, note that the action of the NAD-gobbling CD38 enzyme can be significantly slowed and hindered by CD38 enzyme inhibitors, inhibition that also – like NAD precursor supplementation -- substantially increases cellular stores of NAD. Interestingly, one of the most effective natural inhibitors of CD38 is kuromanin (cyanidin 3-O-glucoside), a plant flavonoid that is found in particularly high concentrations48 in black elderberries like those used to make cold and flu syrups like those sold by Gaia Herbs49 and Sambucol.50 Kuromanin reduces CD38 NAD-consuming enzymatic activity by 50% at a very low 6.3 micro-molar kuromanin concentration.51 There is currently no published information regarding the half-life of kuromanin in the human body, and one researcher52 has cautioned that reducing CD38 enzymatic action using inhibition might have unintended consequences. Nevertheless, plant-derived flavonoid CD38 inhibitors are reported to be non-toxic to humans and actually evidence beneficial effects in animal tests with regard to obesity, heart muscle ischemia, kidney infection, and cancer.53
Go North, Old Man, Go North
It is a commonly held assumption that latitude determines the background vitamin D status in people; i.e., in the northern hemisphere the farther north you live, the more likely it is that you will suffer a wintertime (flu season) vitamin D deficit because of the shorter lengths of wintertime daylight of the northern latitudes. Posting Figure 9 does show that nights are longer (and days shorter) during the late September through early March flu season the farther north that you live. Following this assumption and acknowledging the demonstrated importance of vitamin D levels in minimizing the incidence and severity of respiratory illnesses, it seems a reasonable hypothesis to expect that COVID19 mortality in the US would generally increase the farther north people live.

Posting Figure 10, however, shows that precisely the opposite relationship is true in the United States. The farther north people live in the US, the lower is their general probability of dying of COVID19.
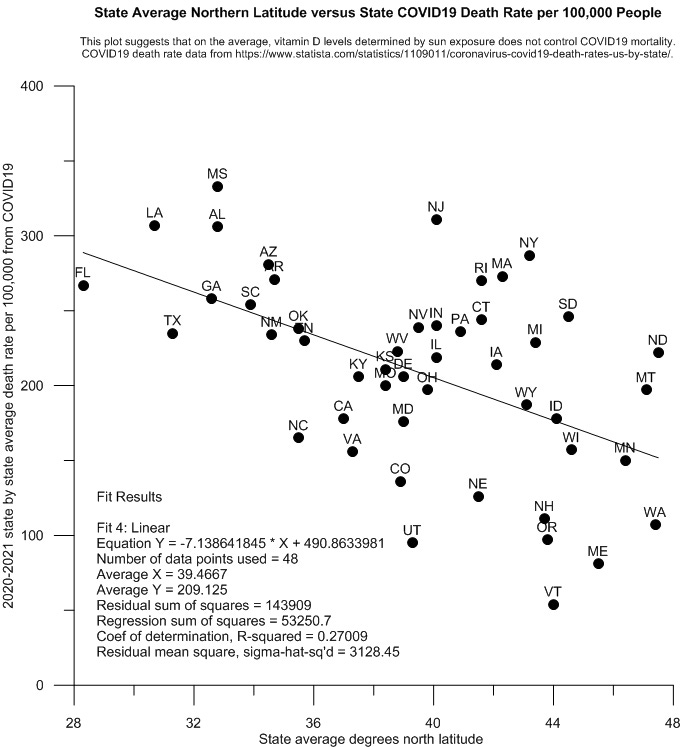
So what gives? Why isn’t increasing latitude and presumed increasing vitamin D deficiency not causing a clear increase in mortality in people living in the increasingly northern states?
My interpretation of this state level data is that increasing northern latitude and longer nights mean that people living in the more northern states are the beneficiaries of increased endogenous generation of the evidently most critical human antioxidant, melatonin, during the respiratory infection season. Length of the nightly melatonin generation by the human pineal gland is controlled by length of the period of night time darkness.54 This means that no matter the age and the consequent relative rate of nightly melatonin production of any given person, if they live farther north, their bodies will benefit from larger nightly doses of melatonin than if they, say, lived in Florida, Texas, or Louisiana instead. On the average, from the map data it appears again that the greater the availability of melatonin to the cells of any given human, the lower is the overall probability that the cell-damaging hyperinflammation associated with severe COVID19 infection will prove fatal.
If you live in the southern hemisphere, this would be changed, of course, to “Go south, old man, go south.”
SARS is a qualitative description of lung injury, while ALI and ARDS are synonyms for the same sort of injury that are also quantitatively defined in terms of extent and degree of injury. See https://www.ncbi.nlm.nih.gov/labs/pmc/articles/PMC7128319/.
https://pubmed.ncbi.nlm.nih.gov/32752010/
https://pubmed.ncbi.nlm.nih.gov/32752010/
IL-6, IL-8, and TNF-α are all pro-inflammatory, cell-signaling cytokines that operate on the outside of nucleus-bearing, tissue and organ-forming cells in the extracellular fluids (https://en.wikipedia.org/wiki/Cytokine). These and other small amino acid chains like them direct the actions of the cell-mediated and antibody-mediated immune systems.
A summary review of the positive and negative aspects of ROS and RNS in mammalian and avian bodies can be found at https://cdn.intechopen.com/pdfs/35982.pdf.
https://www.ncbi.nlm.nih.gov/labs/pmc/articles/PMC5319403/
These degrade rapidly in the gastrointestinal tract, so cannot effectively be augmented orally like dietary antioxidants can. See https://www.hindawi.com/journals/jt/2011/152474/.
https://www.researchgate.net/profile/L-Baroiu/publication/338724315_The_Therapeutic_Relevance_of_Vitamin_E/links/5f6f02f1299bf1b53ef45097/The-Therapeutic-Relevance-of-Vitamin-E.pdf
https://www.researchgate.net/publication/265014760_Theoretical_insight_into_the_antioxidant_properties_of_melatonin_and_derivatives
https://www.ncbi.nlm.nih.gov/labs/pmc/articles/PMC5319403/
https://onlinelibrary.wiley.com/doi/pdfdirect/10.1111/j.1600-079X.2006.00407.x
https://pubmed.ncbi.nlm.nih.gov/12398937/
https://pubmed.ncbi.nlm.nih.gov/17198536/
https://www.frontiersin.org/articles/10.3389/fphar.2014.00151/full
https://pubmed.ncbi.nlm.nih.gov/16316470/
For those who haven’t reached that age yet, various body parts start to get palpably sore and ache on occasion to some degree or another, for no readily apparent reason, in most people by the time they reach the age of 40-45.
Male gender is a predictor of higher mortality in hospitalized adults with COVID-19. Among other things, males don’t get the health benefits of the endogenous antioxidant, estrogen. See https://www.ncbi.nlm.nih.gov/labs/pmc/articles/PMC8270145/ .
https://www.ncbi.nlm.nih.gov/labs/pmc/articles/PMC7228340/
https://pubmed.ncbi.nlm.nih.gov/33680348/
N-Acetylcysteine taken orally increases bodily stores of glutathione. See https://www.ncbi.nlm.nih.gov/labs/pmc/articles/PMC4540061/
These are very high, pharmacological doses of melatonin. Night-time doses of oral melatonin only on the order of slow release 0.3 to 0.5 mg bring the night-time amplitude of plasma melatonin back up to more youthful, but not supra-physiological levels. See https://academic.oup.com/jcem/article/86/10/4727/2849013
https://pubmed.ncbi.nlm.nih.gov/11697548/
https://journals.plos.org/plosone/article?id=10.1371/journal.pone.0239799
https://www.ncbi.nlm.nih.gov/labs/pmc/articles/PMC2912737/
There are a number of factors evidently involved in US African-American average low vitamin D levels besides heightened skin cell protection from UVB light by melanin. See https://www.nejm.org/doi/full/10.1056/NEJMoa1306357 and https://pubmed.ncbi.nlm.nih.gov/31466220/ for more discussion.
https://www.ncbi.nlm.nih.gov/labs/pmc/articles/PMC6780345/
https://www.sciencedirect.com/science/article/pii/S1567576921003222
The formal reduction potential for NAD+ + H+ + 2e- = NADH at pH 7 and 25 C is -0.557 mV (https://www.academia.edu/54383597/NAD_NADH_as_a_model_redox_system_Mechanism_mediation_modification_by_the_environment)
For an excellent seven-minute video explaining the several energy-yielding biochemical reactions/processes/systems involving NAD, see
https://www.ncbi.nlm.nih.gov/labs/pmc/articles/PMC4252787/
https://www.ncbi.nlm.nih.gov/labs/pmc/articles/PMC7322475/
https://link.springer.com/article/10.1007/s10557-020-07007-8 and https://www.researchgate.net/publication/13490277_Analysis_of_the_distribution_of_human_CD38_and_of_its_ligand_CD31_in_normal_tissues
https://www.ncbi.nlm.nih.gov/labs/pmc/articles/PMC5942225/ Some recent reports suggest the CD38 enzyme is actively spread throughout the human body over time via the agency and movement of various white blood cells. See https://www.nature.com/articles/s42255-020-00298-z and https://www.nature.com/articles/s42255-020-00305-3.
Another factor in this decline is declining chemical activity of another enzyme, NAMPT, that helps recycle waste product NAM produced by the SIRTS, PARPS, and CD38 enzymes back into NAD. Decline in the chemical activity of NAMPT is associated with age, obesity, chronic inflammation, and physical inactivity.
https://pubmed.ncbi.nlm.nih.gov/33051211/
https://pubmed.ncbi.nlm.nih.gov/32386566/ and https://www.ncbi.nlm.nih.gov/labs/pmc/articles/PMC6342515/
https://www.amazon.com/Rugby-Niacin-500-1000-Tabs
https://pubmed.ncbi.nlm.nih.gov/32386566/
http://phenol-explorer.eu/contents/polyphenol/9?utf8=✓&options%5Blist_format%5D=list&options%5Bsort_by%5D=by_average&options%5Bempty_rows%5D=off&options%5Bunit_type%5D=weight
https://www.amazon.com/Gaia-Herbs-Black-Elderberry-Syrup . Note that the black elderberry syrup made by Gaia Herbs is almost 8xs more concentrated than that made by Sambucol. Assuming no gastrointestinal, liver pass-through, or other loss of the kuromanin in fresh black elderberries, a back-of-the-envelope calculation indicates that the recommended syrup dosages provided by both Sambucol and Gaia Herbs should at least temporarily bring blood plasma kuromanin concentrations into the near neighborhood of 6.3 micro-molar concentration that would reportedly reduce CD38 activity by about 50%. This estimate was made for a 200 pound man taking the recommended daily adult dose of each of the syrups.
https://www.amazon.com/Sambucol-Elderberry-Original-Formula-Antioxidant
https://pubmed.ncbi.nlm.nih.gov/21641214/
https://pubmed.ncbi.nlm.nih.gov/19149603/
https://www.frontiersin.org/articles/10.3389/fimmu.2019.01187/full
https://en.wikipedia.org/wiki/Melatonin